Note: finance topics are slightly sensitive, so, while nothing in this article contains proprietary information, I will not include the names of people I talked with for this piece. I appreciate everyone who reached out to help me put this together!
Finance tries to make failure survivable: the Andrew Lo thesis
Finance makes future success tradable: royalties and synthetic royalties
Conclusion: what does finance teach biotech to value, and should we worry?
Introduction
For $250 million and ten years of your life, you may purchase a lottery ticket. The ticket has a 5% chance of paying out. When it does pay out, it pays roughly $5 billion. A quick calculation will show you that the expected value of the ticket is $250 million. This is essentially what drug development is. Or rather, it’s what drug development was, twenty years ago. The upfront payments have been climbing, the hit rates falling, and expected values have, at best, held flat. Should you buy a ticket?
Perhaps not. In fact, any reasonable player should have long since stopped playing this stupid game. Unfortunately, we still need drugs. People have cancer, and heart failure, and Alzheimer’s, and a thousand genetic diseases that nobody has ever heard of, and the only industry on Earth currently set up to do anything about any of this is the same industry running the lottery-ticket business described above. The game is dumb and we need it played anyway.
So the real question is not whether to play, but how to make playing less awful for this involved. And the answer, increasingly, is ‘financial engineering’: a set of structural tricks that let people hold more tickets than they otherwise could, or buy a fraction of the winning tickets after they’ve been drawn, or some other strange, clever thing that all financiers find obvious and everyone else has never heard of. All this, done to trade and barter over the risk inherent to the whole enterprise, slicing it into pieces small enough that someone, somewhere, is willing to hold each one in exchange for something.
I’ll walk through a handful of these, the people who invented them, and case studies involving the tactic. And at the end, we’ll ask the question of whether all these tricks are, in aggregate, altering what the pharmaceutical industry decides to value.
The first such trick, and the one that perhaps kicked off the start of the whole effort, was dreamed up by a man named Andrew Lo.
Finance tries to make failure survivable: the Andrew Lo thesis
Andrew Lo is a finance professor at MIT's Sloan School of Management. Among all TED talks that have ever been produced, there are few worth watching. Andrew’s talk, which has the wonderful title ‘Can Financial Engineering Cure Cancer?’, is one of them:
I recommend you listen to the full thing, because it really is quite good. If you’re strapped for time, the core thesis is as follows:
Individual drug programs fail about 95% of the time. But this doesn’t mean the expected value of a single program is necessarily bad. As I said at the start: a 5% shot at a $5 billion blockbuster against a $200 million development cost is technically positive EV on paper. But this implies that you need to be able to survive the 95% of outcomes in which you lose everything, and most investors, reasonably, will not.
Lo's insight, published in a 2012 Nature paper, was simple. Just bundle 50 or so drug programs into a single entity, one with a war chest of $5 to $15 billion, and roll the dice. The individual drug programs are still terrible standalone bets, but if they're sufficiently uncorrelated, at least one is almost guaranteed to hit, and it will hit big enough to pay off all the programs that failed. Which means you can keep playing, forever. Of course, the ‘uncorrelated’ bit is the ‘spherical cow’ part of all this. It’s impossible to do it perfectly, but it can be done well enough for risk to fall dramatically.
There’s an extra layer of complexity here about how if you can get the portfolio risk to be low enough, you can issue debt against the portfolio to sell as bonds, which unlocks a much larger pool of non-venture capital who want more stable returns. This is arguably the most interesting thing that Andrew believed in, but this particular bit never really went anywhere. We’ll discuss the obvious ‘why not?’ question at the end of this section.
The direct descendant of this whole thesis—at least the ‘drug portfolio’ part—is BridgeBio Pharma, founded in 2015 by Neil Kumar, who was Andrew’s student at MIT. It is structured almost identically to Andrew’s original thesis: a central holding company that creates subsidiary companies, each focused on a single rare disease. Each subsidiary has its own equity structure, its own management team, and 1-2 drug programs. If a subsidiary's drug fails, it dies, but BridgeBio survives. If it succeeds, the parent holds enough equity to capture massive upside. The company IPO’d in 2019, is now worth billions, and has a pretty good stock trend for a biotech.
There are spiritual cousins as well, such as Roivant Sciences, founded in 2014 by Vivek Ramaswamy. It has a nearly identical corporate structure to BridgeBio—what’s come to be known as a ‘hub-and-spoke’ model—but whereas BridgeBio does de novo drug development in rare diseases, Roivant in-licenses drugs that big pharma has abandoned for non-scientific reasons: portfolio reprioritization, executive turnover, M&A reshuffling, quarterly earnings pressure. There are lots of these molecules floating around, and if you hire good enough people, you have the ability to spot them before anyone else. Roivant went public in 2021 at a $7.3 billion valuation, and its subsidiaries have completed twelve consecutive positive Phase 3 studies. And it has an even better stock history!
This solves the fundamental problem of biotech, no? Really, in retrospect, it’s astonishing that we let anybody create a non-hub-and-spoke biotech. You have a set of bets, each one of which is individually stupid, and then you put them in a bag, and the bag becomes smart by virtue of each bet being insanely high variance. It is the obvious thing to do.
Unfortunately, upon trying this out, we will run into two big problems. The first one is that running many drug programs at the same time is really hard. And the second one is that people know running many drug programs at the same time is really hard, and they will price any attempt to do so accordingly.
An exemplar of the first lesson is Centessa Pharmaceuticals. Centessa was founded in late 2020 by Medicxi, a life-sciences venture firm, as another implementation of this thesis: ten private biotech companies, each with its own single asset, combined under one holding entity, taken public in May 2021 at $20 a share. Though they are often held up as paragons of the Andrew Lo thesis (including by me!), Roivant and BridgeBio weren’t real hub-and-spoke enthusiasts. Centessa was. Whereas Roivant in-licensed abandoned pharma assets and BridgeBio concentrated almost entirely on rare genetic disease, Centessa bravely stuck to the Lo script: a portfolio of genuinely uncorrelated clinical risk. Their spokes covered: hemophilia, oncology, pulmonary hypertension, narcolepsy, fibrotic disease, autoimmune disease — if there was any correlation risk, it was that drug development was occurring at all.
The model did not work. Within eighteen months Centessa was shutting down spokes. By 2023, they had abandoned the hub-and-spoke model entirely and pivoted to a single-asset company focused on orexin agonists for sleep disorders. That pivot, to be clear, worked spectacularly. Lilly bought them for $6.3 billion in early 2026, making Centessa one of the more successful biotech exits of the decade. But they got there by becoming a single-asset company. What had gone so wrong with the original thesis? The surface answer is a mix of capital and luck. Several spokes failed on their own merits, and the 2022-ish biotech market crash closed the door on funding whatever was left. Centessa shareholders ended up all right in the end, but hub-and-spoke models are empirically not silver bullets for the hard problem of drug development.
The second problem here is that people simply may not believe in your so-called ‘uncorrelated risk portfolio’. This will obviously happen when you raise money to pursue the venture, and it will, surprisingly, happen again once you go public.
As an example: did you notice that big drop in BridgeBio’s stock in late-2021? This is when their lead candidate acoramidis—a treatment for a rare heart condition called transthyretin amyloid cardiomyopathy—failed to beat placebo on its primary endpoint in a Phase 3 trial. The stock dropped 72% in a single day. This was not the tidy portfolio-theory response. The rational response would be “well, BridgeBio has four other clinical-stage programs and $800 million in cash, so the diversified portfolio thesis should protect us." The market said "holy shit, the lead asset is dead, the portfolio theory behind this company is nonsense, sell it," and priced that sentiment accordingly.
The funny part of this all is that BridgeBio kept running the trial. The 12-month primary endpoint had failed, but the study was designed to run to 30 months, with a harder secondary endpoint: death and cardiovascular hospitalization. In July 2023, the longer-term data read out, and acoramidis worked, with the secondary endpoint being met. The stock surged 76% in a day, BridgeBio eventually won FDA approval, and the drug—now on the market—is called Attruby. Stressful!
Well, that’s that. But we should return to Andrew Lo for a second. The part of Lo’s idea that did not arrive, at least not in its original form, was the bond-market part. Why has no one implemented what was arguably the most clever part of his pitch: issuing debt against your drug portfolio, allowing you to access vast sums of institutional, low-risk capital?
Well, to some degree, someone has, but only for approved drugs. BioPharma Credit is one such institution, and makes secured loans to commercial-stage biotechs, typically collateralized by the revenue stream of one or more approved products.
But nothing like this exists for clinical-stage stuff. Why not? Happily, Lo himself offered an answer, almost a decade after his first paper. For one, biotech is simply not used to that type of financing so they don’t do it, and two, the extreme scale of financing that this unlocks has simply not yet been needed, so nobody can raise it. But the third most important point is a lack of institutional support. There is no biomedical Moody's—no quantitative, authoritative voice that can tell a pension fund how risky a portfolio of drug assets is. And even if there were, there is no biomedical Fannie Mae—no government-backed entity that acquires biopharma loans and securitizes them into something an institutional allocator would actually buy. Our field exists in the same state that mortgages were in the 1930s, which were considered too risky for banks to buy until the federal government created these two pieces of infrastructure to make it safe.
But, Lo posits, the need for capital eventually changes behaviors, biology is poised to only grow far larger than it is today, and models for drug portfolio risk adjustment are only getting better. Four years after the paper, I am unsure whether much has changed, but we’ll see what the future holds.
Finance makes future success tradable: royalties and synthetic royalties
Drug royalties are pretty simple. You discover an interesting target or chemical, but don’t want to bother with developing it further. So you pawn it off to a big pharmaceutical company with a lot of resources, alongside a contractual agreement that you’ll receive 3% of net sales if a drug based off your work is eventually approved and commercialized. And like any contractual agreement, it can be bought and sold.
Royalty Pharma, founded in 1996 by Pablo Legorreta, is the largest company in this market and possibly the purest expression of financialized drug development that exists. It has no labs, no therapeutics arm, and no ambition to discover drugs itself. It buys royalties, from universities, academic medical centers, small biotechs, individual inventors, and holds them. The portfolio includes claims on 7 of the 30 most-prescribed drugs in the United States. It reported $2.38 billion in revenue in 2025 from what is, spiritually, a filing cabinet.
Is this rent-seeking? If you look at the details, it actually feels pretty fair to all parties involved. A university that has a royalty over some particular drug developed by a professor has no ability—or desire!—to forecast its chance for success, its revenue if approved, or how to hedge the risk that a competitor enters the market. It also very likely has a preference for less money today than more money over the ten years of a drug’s exclusivity period. Royalty Pharma and its competitors have the opposite preferences and all the abilities the university lacks. The university gets liquidity and certainty; Royalty Pharma gets a claim on an approved drug's revenue stream at a discount to its expected value. Both sides win.
But the more interesting recent development is the rise of synthetic royalties.
A traditional royalty is a pre-existing legal right. It exists because someone did the original research and negotiated a licensing agreement. A synthetic royalty is different. It’s a manufactured financial claim on future drug revenues that didn’t previously exist. Consider an example: a biotech company has a drug in clinical development, one that it owns entirely. It needs money. It doesn’t want to issue equity (dilutive) or take on debt (requires collateral). So it invents a drug royalty from scratch, an entirely new obligation that did not previously exist, and sells that. Now they do not own the drug’s IP entirely, some other party owns 3% of the future sales of it if it ever succeeds, and the biotech gets non-dilutive capital today.
What’s the difference between these increasingly complex synthetic royalty agreements and typical, bespoke pharma deals? They feel similar. And yes, they are functionally equivalent in terms of cash flow or deal structure. Where the difference lies is in each party’s intent. In typical pharma deals, the buyer cares about something strategic, say, operational control over a drug’s development journey. Buyers of royalties, synthetic or otherwise, do not care about that. They care entirely about the probability-weighted present value of the future payments, and you can imagine how useful this decoupling of capital from often burdensome partnership demands can be.
The royalty market is, in some sense, a secondary market for the financial value typically embedded in pharma licensing agreements. And it's still early.
One report found that there were 102 major royalty transactions from 2020 to 2024, noting that synthetic royalties are growing at 33% annually. The buyer pool includes not only royalty-centric funds like Royalty Pharma, but increasingly pension funds and private equity as well. The same institutions Andrew Lo wanted to be in on biotech are getting in on the game, just in a different way.
This whole class of synthetic royalties is growing more complex over time, with some even including milestones built into the sold contract, such that the seller (the biotech) receives even more upfront capital upon the achievement of Phase 2 trial success or outright drug approval. The whole concept is also growing physically larger. In June 2025, Royalty Pharma and Revolution Medicines announced a $2 billion funding agreement—$1.25 billion of which was structured as a synthetic royalty—to fund the development of daraxonrasib; the largest ever transaction in this particular asset class.
But at the same time, within synthetic royalties, you can see the beginnings of a financial instrument that is strange enough that one cannot easily predict its second- or third-order effects. Pharma partnership agreements can be burdensome in the demands they make, but they are at least ‘time-bounded’ in ways that are easy to plan for. Synthetic royalties follow a company around forever, as long as a drug is under patent, actively extracting value all the way, their only contribution being an initial surge of capital. This is nobody’s fault of course, least of all the royalty holders. ‘We are selling to willing buyers at the current fair market price’ and all. But the cumulative effect, as more drugs carry more synthetic royalty obligations, is a pharmaceutical economy where an increasingly large fraction of every dollar of drug revenue is pre-committed to financial intermediaries before the drug reaches a single patient.
But it’s not as cut and dry as ‘synthetic royalties are bad' because of this. Consider the Revolution Medicines case from earlier. Their drug daraxonrasib has a strong chance of being a blockbuster, and so scaling global commercialization will be enormously expensive. An equity raise would have diluted ownership right before value-inflecting Phase 3 readouts (which were excellent!), traditional debt at that scale would be impractical, and a pharma partnership would surrender commercial rights to what could be a decade-long franchise of label expansions. The synthetic royalty allowed Revolution to sidestep all three, largely as a result of the royalty being tiered, decreasing with sales volume, and dropping to zero above $8 billion in annual net sales. If daraxonrasib becomes a true blockbuster, the royalty burden effectively caps out and becomes negligible as a percentage of revenue.
But why would Royalty Pharma agree to this at all? Isn’t this clearly not in their favor? Not at all: they likely just did the numbers, and anything above some certain threshold in yearly sales is both unlikely and unneeded for their portfolio math, so they are happy to give the tail scenario away for free.
All of this, only possible because there is an entity willing to buy a manufactured claim on future revenue that didn't exist until someone decided to create it. The royalty market shows the basic pattern: once a future drug cash flow becomes legible, someone will turn it into a security.
Finance rewrites the incentives: PRVs and CVRs
What we’ve discussed so far assumes some degree of intentionality. Andrew Lo purposefully came up with his thesis, Royalty Pharma deliberately built a business around drug royalties, and so on. But there are two particular financial instruments that were intentionally designed at the start, but have slowly begun to display an unpredictable life of their own once deployed. I’d like to discuss them because I think they do a great job in demonstrating not only how tradable instruments in finance can have material impact in how drug development works, but also how those impacts can be very difficult to predict in advance.
The two are PRVs, or Priority Review Vouchers, and CVRs, or Contingent Value Rights.
We’ll start with PRVs.
In 2006, three professors at Duke published a paper titled “Developing Drugs for Developing Countries". In it, they discuss a well-trodden problem: infectious and parasitic diseases create enormous health burdens in the developing world, but because the people suffering from them are poor, there's essentially no commercial incentive to develop treatments. Of course, ideally there would be some way to incentivize for-profit companies to do it. But financial incentives require money, and money requires Congress, and Congress requires political will that rarely materializes for diseases affecting people who can't vote in U.S. elections.
The fix, the authors argued, is to use a logistical incentive instead. If you are willing to develop a drug for a neglected disease, the government ought to help you out somewhere else in your drug portfolio.
How? By offering you a PRV. But what use is the PRV? Once a pharmaceutical company has wrapped up their clinical trial work and submits an application to the FDA for official approval, they must wait 10 months for FDA review. But if they submit this one-time-use-only voucher alongside the application, FDA should be forced to give you a review within 6 months. And just in case you don’t actually have an internal portfolio of drugs to allocate this PRV to, the voucher should also be sellable. Four months of time-value of earlier market entry for a ‘top-decile’ drug can be worth an awful lot, around $300 million according to the authors.
You can imagine a very neat feedback loop from all this. For instance, a small nonprofit or academic group develops a river blindness treatment, receives a voucher, and can then sell the voucher to Pfizer to use the proceeds to fund more neglected disease work.
In a rather astonishing act of ‘listening to healthcare economists’ that I don’t believe ever occurred thereafter, Congress enacted the program in 2007, just a year after the paper’s publication. It expanded again in 2012 to include rare pediatric diseases. And again in 2016 to include medical countermeasures against biological/chemical/radiological threats.
There are two things I find very interesting about PRVs.
The first is that, as the title of this section implies, the PRVs have gained secondary market price dynamics that its creators never intended. The buying cost of a PRV at any given moment is a function of how many are floating around, how many blockbuster drugs are approaching FDA submission, the competitive landscape, and whether Congress has recently done something to expand or contract the program. AbbVie paid $350 million for a single voucher in 2015—the all-time high, driven by the voucher being the only one out there and that their competitor was releasing a similar drug to theirs. Novartis picked one up in 2023 for $21 million—the all-time low.
How did Novartis get one for so cheap? Funnily, that particular story also illustrates the increasingly complex financialization of biotech quite well. When Novartis licensed a particular drug to a particular biotech back in 2019, it baked in a “pre-agreed, contractually defined percentage of the PRV value” into the licensing agreement four years before the voucher existed, and, in fact, before the drug itself had even been approved. And when the biotech got the drug approved and received the voucher in 2023, Novartis simply exercised the option to purchase it for a ridiculously low value.
Imagine being the person behind that deal!
The second thing, even more interesting is that the whole program has increasingly begun to bear no fixed relationship to the social good it was meant to incentivize. Why? Because even at the voucher's peak secondary market value of $350 million—though it usually oscillated around the $100M mark—it was not enough to shift a large pharma company's portfolio allocation in any meaningful way. In the few cases it did, it shifted it towards doing the absolute, most bare-minimum possible thing: approval of the drug, not utility of the drug. The voucher pays for the regulatory event, not the public-health outcome. In a great paper titled ‘The priority review voucher: a misconceived quid pro quo’, the authors say this:
…the PRV, except few examples, has largely failed to deliver medical benefits for patients suffering from neglected diseases because it rewards obtaining FDA marketing authorisation without regard for the products actually being available, affordable and equitably accessible for people.
Now, it would be lying to tell you that PRVs have not helped anyone. They have! But there have been enough cases of bad behavior here that it is worth wondering if there is something better that is possible. This is, in fact, being worked on, but it takes us off topic, so I’ve put some details about it in the footnotes1.
The second financial instrument I want to discuss is the CVR, or Contingent Value Right.
CVRs are simple. When an acquirer and a target company cannot agree on what a drug-in-development is worth—which is most of the time—they structure a simple conditional payment. If the acquired drug(s) hits a specified milestone, the acquirer pays the target's former shareholders an additional sum. Most CVRs are structured like normal pharma partnerships, as in, a closed, non-transferable contract between two partners. A small minority of them are structured as tradable securities, listed on the NYSE or Nasdaq with their own ticker symbols, but these aren’t particularly special beyond their raw size.
What is most interesting about CVRs is the perverse incentives they sometimes create.
When Sanofi acquired Genzyme for $20 billion in 2011, Sanofi issued CVRs tied to the regulatory approval and commercial success of Lemtrada (alemtuzumab), an MS drug that Genzyme had been developing. Up to $3.8 billion was on the table if the drug hit its milestones. But Sanofi was also simultaneously developing its own MS drug, Aubagio. Aubagio had no CVR obligations attached to it.
Sanofi was now contractually obligated to compete vigorously against itself, on behalf of strangers, for free. Predictably, it did not.
Obviously, Sanofi was sued for this. The former shareholders alleged that Sanofi deliberately slow-walked Lemtrada's FDA submission and under-invested in its commercialization to minimize CVR payouts. But deliberate sabotage is hard to distinguish from ordinary sluggishness. Sanofi settled in 2019 for $315 million—well short of the $708 million in missed payouts the shareholders claimed—without admitting wrongdoing.
The pattern repeated more recently and at even larger scales in 2019, with BMS's $74 billion acquisition of Celgene, in which $6.4 billion in CVR payouts hinged on three drugs hitting FDA approval by fixed deadlines. Two were approved on time. The third missed by thirty-six days. As a result, the entire CVR expired worthless. As you may expect, former shareholders again sued.
If we were to generalize this, the structural problem is simple: a CVR can make the buyer responsible for creating a payout that the buyer would rather not pay. But if that’s the case, why are CVRs—which are accelerating in their popularity—done at all? For one, the above case studies are very much not the norm, most go on perfectly fine. And two, the value of CVRs as a coordination mechanism, even when they go wrong, empirically outweighs the later headaches they cause.
Finance reaches failure itself: zombie biotechs
Royalty Pharma is not the only player in the royalty space. There are a few others, one of them named XOMA Royalty. XOMA is especially interesting, because it was once a traditional biotech company that developed and licensed drugs. And in 2017, it pivoted to become a royalty aggregator. And starting in 2024, it began to poke at the business of buying up, and liquidating, ‘zombie biotechs’.
Zombie biotechs are publicly traded companies whose stock price is below the cash on the balance sheet. This translates to investors saying that their IP, patents, clinical data, team, all of it, is not only worthless but is actively destroying value by burning through cash that would be better deployed sitting underneath a bed. Roughly 300 companies fit this description in mid-2024, most of them casualties of the 2020-2021 IPO bubble, when a lot of biotechs went public that had no business doing so.
These companies can’t raise equity (who would buy?), can’t take on debt (against what collateral?), and can’t be bought/merged with anyone (who would want them?). In an ideal world, the founders would simply put the whole business out of its misery, but they are collecting a paycheck anyway with their dwindling cash reserves and closing down a public company is a surprisingly legally fraught thing to do. So they just wander around as zombies.
XOMA’s insight was that this particular purgatory may itself be an asset class. They step in, acquire the company at or below cash value, and return cash to the shareholders who have been trapped in a slowly deflating stock for years. Then, they take a close look at everything the company created—patents, clinical data packages, licensing rights, partially completed regulatory filings—and sell it, keeping the profits for themselves. Or simply hold it, just in case it’ll be useful elsewhere in their portfolio.
The concept itself is not new. This is vulture investing, translated into biotech. But whereas a typical vulture investor’s goal is to flip an entire company onto someone else, the biotech vulture capitalist’s hope is to sell off pieces of the company. And the pieces can be surprisingly valuable. A drug candidate that failed a Phase 2 trial in one indication can be worth millions to, say, some of the hub-and-spoke companies we discussed earlier. Maybe Roivant believes that the endpoint was misspecified, or the indication was wrong, or that the drug is indeed useless, but that the PK/PD, safety signals, biomarker responses, regulatory responses, and dose-response curves uncovered during the trial are useful, and they’d be willing to pay vast sums for that data. What XOMA does here is make this information legible to potential buyers.
To help illustrate this, let’s consider a case study: Kinnate Biopharma.
Kinnate was an oncology company developing kinase inhibitors for cancer patients with specific genetic mutations. As the story goes for many companies of that era, they went public and by early 2024 were trading below their cash balance. There was no outright clinical trial failure, they simply ran out of money to develop their drugs further. In February 2024, XOMA announced it would acquire Kinnate for roughly $2.50 per share in cash, or $126 million. Then, over the next year, XOMA sold all five of Kinnate’s pipeline assets to other companies. In April 2025, they announced the completion of these sales, with terms entitling XOMA to up to $270 million in upfront and milestone payments, plus, keeping to their name, ongoing royalties ranging from low single digits to mid-teens on commercial sales. Kinnate’s shareholders received most of the upfront payment, and XOMA got to double its money in flipping the assets.
What would the counterfactual be if XOMA had not stepped in? Kinnate would’ve continued to bleed cash until it ran out. At that point, the IP would have been worth even less—the utility of biological information depreciates fast!—and the shareholders would have gotten back even less, perhaps nothing at all.
There is another player in this space worth discussing: Kevin Tang, through Tang Capital and its acquisition vehicle Concentra Biosciences. By mid-2025, Concentra had become one of the busiest buyers in biotech, making repeated bids for distressed public companies, with the explicit intention of closing them down, selling whatever assets could still be sold, returning some cash to shareholders, and keeping whatever spread remained.
Isn’t this quite similar to XOMA? Yes, both XOMA and Concentra are buyers of distressed, sometimes very clearly, biotechs. But the difference is when they arrive. XOMA typically shows up at the doorstep of companies that are clearly on death's door. But Concentra often arrives earlier, while the public company is technically alive and its board is still weighing bad alternatives: reverse merger, dilutive financing, slow wind-down, strategic review, or sale. And Concentra aggressively attempts to force the boards hand into a sale to them.
To be fair, ‘force’ is a bit strong of a word here. A better term would be ‘an offer they can’t (easily) refuse’. Concentra’s pattern is to accumulate a large minority stake, make an unsolicited bid, and dare the board to explain why shareholders should keep funding the burn instead of taking cash now.
Why can’t they refuse it?
Consider Jounce Therapeutics. In February 2023, Jounce announced a reverse merger with Redx Pharma, alongside a 57% workforce reduction. This was not exactly a happy ending, but it was at least a biotech ending: Redx’s pipeline would become the core of the combined company, Jounce shareholders would own a minority stake, and some version of the organization would continue to exist. Then Concentra appeared with an offer that promised even more liquidation to the shareholders, but one that would completely strip-mine Jounce to sell off as parts.
Tang is not doing anything illegal here, nor are boards literally compelled to accept every higher bid that comes along. But once a company has put itself in sale mode, the board starts to look less like a steward of a scientific project and more like an auctioneer for whatever value remains. This creates a bleak asymmetry. A reverse merger can be better for the people inside the company, better for the local biotech ecosystem, and perhaps even better for the vague moral category of “letting the science continue.” But that is not the job of the board to further. Their job is to ensure the shareholders are best served, and for them, Concentra’s highly liquid offer is difficult to argue against. In Jounce’s case, the Concentra transaction also came with an 84% workforce reduction. The board went with the Concentra offer.
Curiously, there are ways for companies to fight back against Concentra, and fight back they have. Their weapon is colloquially referred to as a ‘poison pill’, and goes as follows: if Tang keeps buying shares and crosses a threshold, usually around 10%, then every other shareholder receives the right to buy more stock at a discount, instantly diluting Tang. This does not resurrect the company, and it does not make Tang go away. It simply prevents Tang from buying enough stock in the open market to make liquidation feel inevitable before the board has themselves decided it is inevitable.
This is all quite interesting. But it is likely a transient phenomenon. The zombie biotech liquidation market is a finite resource; the 300 companies trading below cash are overwhelmingly a product of the 2020-2021 vintage, a specific historical moment when the bar for going public was unusually low. That cohort is being worked through. Some will be acquired by the players discussed here. Others will manage to raise capital and survive. Most will simply wind down on their own, returning whatever cash remains to shareholders without the intermediation of a vulture buyer. Unless there’s another IPO bubble of comparable scale soon, the supply of zombie biotechs will shrink over the next few years, and the opportunity that is currently being exploited will narrow.
So why do I mention this at all?
The zombie biotech business is worth dwelling on because it marks a kind of endpoint. Whereas every other instrument in this essay financializes drugs that might still become therapies, these are different. XOMA financializes drugs that won’t, and Concentra financializes drugs that likely won’t. If the frontier of financial creativity has reached the dead and dying, it tells you something about how thoroughly every other surface has already been colonized.
Conclusion: what does finance teach biotech to value, and should we worry?
Andrew Lo’s original insight was not that finance could make drug development easy. Nothing can make drug development easy. His insight was that finance might make failure survivable. I think this is directionally correct. Financialization is just the process of making implicit economic relationships explicit and tradable, and more liquid markets for biotech risk are almost certainly better than fewer. And what has happened since Lo’s 2012 paper is that financial engineering has been applied not just to the drug portfolio problem, but to every conceivable surface of the drug development process: partnerships, mergers, royalties, and even the death of a company.
Is this a bad thing? Probably not. Objecting to the decoupling of finance from therapeutic value is a bit sentimental, since, in theory, financial incentives should track therapeutic value. But how confident are we about that? Are we boiling a frog here? And if we are, what exactly is the frog?
Like I said at the start, it is important to understand that financial engineering is happening for a reason: this whole industry is excruciatingly difficult to build something in. It’s only getting worse too. Starved of capital, clever people will figure out ways to offer it in increasingly exotic forms to increasingly desperate scientists or companies, and little can prevent these two from finding each other at a bar. The alternative to a financialized biotech industry is not some prelapsarian era of pure scientific inquiry. It is the same industry, with the same problems, but less money and fewer ways to deploy it.
But let’s say we are being idealistic here. What, then, should worry us about financialization squishing its way deeper into drug development? I’m happy to raise my hand first: I'm a little worried about whatever private-equity did to hospitals happening, in slower and less visible ways, to molecules themselves. Yes, I realize the nature of drug discovery imposes a constraint that most financialized industries don't have: the thing has to actually work. The FDA is a binary filter that no amount of financial engineering can route around, and as long as that's true, the typical finance-driven enshittification story shouldn’t apply here.
But "working" and "mattering" are not the same thing. For instance, you’ll notice that both Roivant’s and BridgeBio’s drug pipelines share a similarity: a focus on rare diseases. Finance people love rare diseases. Small trials, clear genetic etiology, often no existing standard of care, accelerated approval pathways, and excellent unit economics. This is fantastic for the several hundred, perhaps several thousand, patients helped by this work, and I don’t intend to minimize it. But would GLP-1s come out of this process? Would PrEP?
This doesn’t have to be a big deal. All of these could coexist. Big pharma and startups continue to have high variance bets, the financialized folks stay low variance, they work together when needed, the world is at peace. But capital is finite, and drug development keeps getting more expensive and less predictable. My worry is not that BridgeBio and Royalty Pharma are doing something bad. They aren’t, and are in fact doing something very good. The worry is that they are doing something so legible, so well-suited to the preferences of the capital markets, that the money increasingly, naturally flows to them and nowhere else.
Is this a real worry?
On one hand: obviously not. The sort of financialized rare disease work presented here may look quite good, but it still makes up an extremely small portion of biotech funding—around 2%. And it is not like Roivant or BridgeBio are poking at some genuinely undiscovered alpha. They are about a decade old, and despite their success, still don’t have many peers. Maybe this market is self-limiting. Maybe there are only so many BridgeBio-shaped opportunities in the world, and the rest of the biotech-earmarked dollars must go towards the higher-variance stuff.
On the other hand, the counterargument is the patent cliff. Between 2025 and 2030, patents for nearly 200 drugs are set to expire, including roughly 70 blockbusters. More than $300 billion in revenue is at risk, or about one-sixth of the industry’s annual revenue. Patent cliffs are normal, but this one is unusually large, weighing in at three times the size of the cliffs of the 2010s in lost revenue. Five of the top 10 pharmaceutical firms face a potential hit exceeding 50% of their current revenue.
What changes after an event like that? Perhaps Big Pharma will increasingly look towards easier, lower-risk/lower-reward diseases. Maybe they’ll be increasingly sympathetic to royalty and synthetic royalty funding agreements, further cutting into the economics of a drug. Maybe this leaks over into the public markets, and the diffuse preferences of a thousand allocators would rather fund the pharmaceutical companies who go down that path, instead of continue with the status quo.
The frog is not any single drug or company. It is the industry’s willingness to fund biology that is illegible, expensive, and likely to fail, which is to say, the kind that occasionally changes the world. Again: financial engineering did not create this problem—that fault can be attributed entirely to R&D productivity decline. In fact, the financiers may even be an especially brave vanguard in giving biotech the veneer of being a viable asset class. But they still may wind up making the response to the underlying problem worse by offering a way to achieve returns in ways that slowly diminish our institutional capacity to create the next generation of revolutionary medicines.
To end this off: I have deliberately left out China, which may be the most aggressive current example of financial architecture shaping a drug pipeline. That deserves its own essay, and will get one soon.
In June 2025, the CNPV (Commissioner’s National Priority Voucher) was announced by FDA Commissioner Makary, and represents a brave new direction of the concept: a non-transferable voucher that can be used for a 1-2 month review period and is awarded based on alignment with “critical U.S. national health priorities.” What does this mean? Nobody knows!
What we do know is that 18 vouchers have been awarded so far, 4 products have been approved through the program, and the whole thing has basically zero external visibility. If you go online, there is a lot of distaste about the whole thing, including two lawmakers who expressed that the program could “enable corruption by creating a new, lucrative gift for drugmakers and allies politically favored by President Trump.” I get it. But I think there is actually some utility in drug approval processes that are bespoke enough to let the federal government both accommodate practical constraints—manufacturing limitations, supply chain fragility—and extract concessions like price adjustments in return for regulatory speed. Obviously not ideal that such a program exists in the context of the volatile current administration, but I’m not especially opposed to a ‘we’ll fast-track good stuff through an opaque review process’ setup.



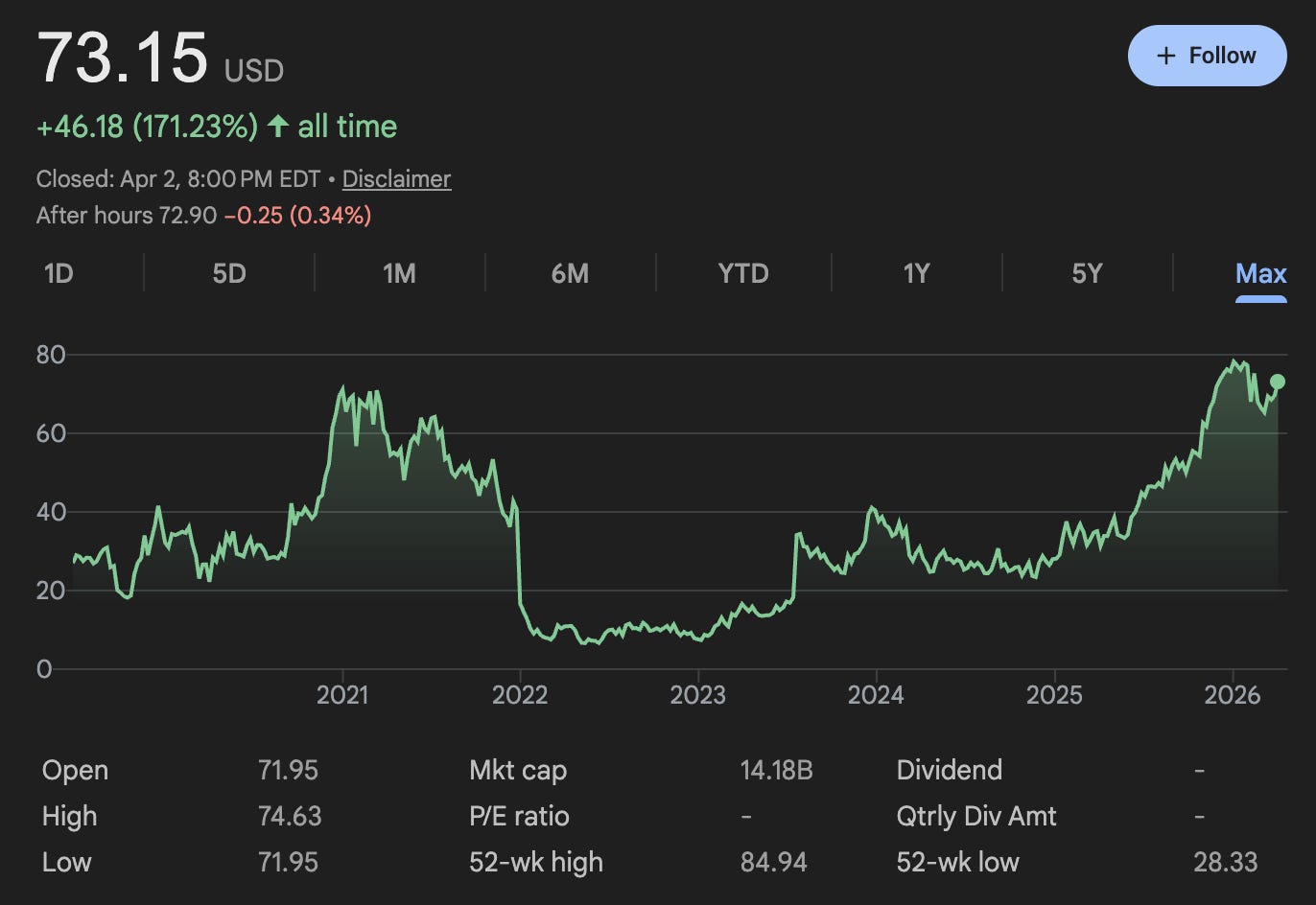
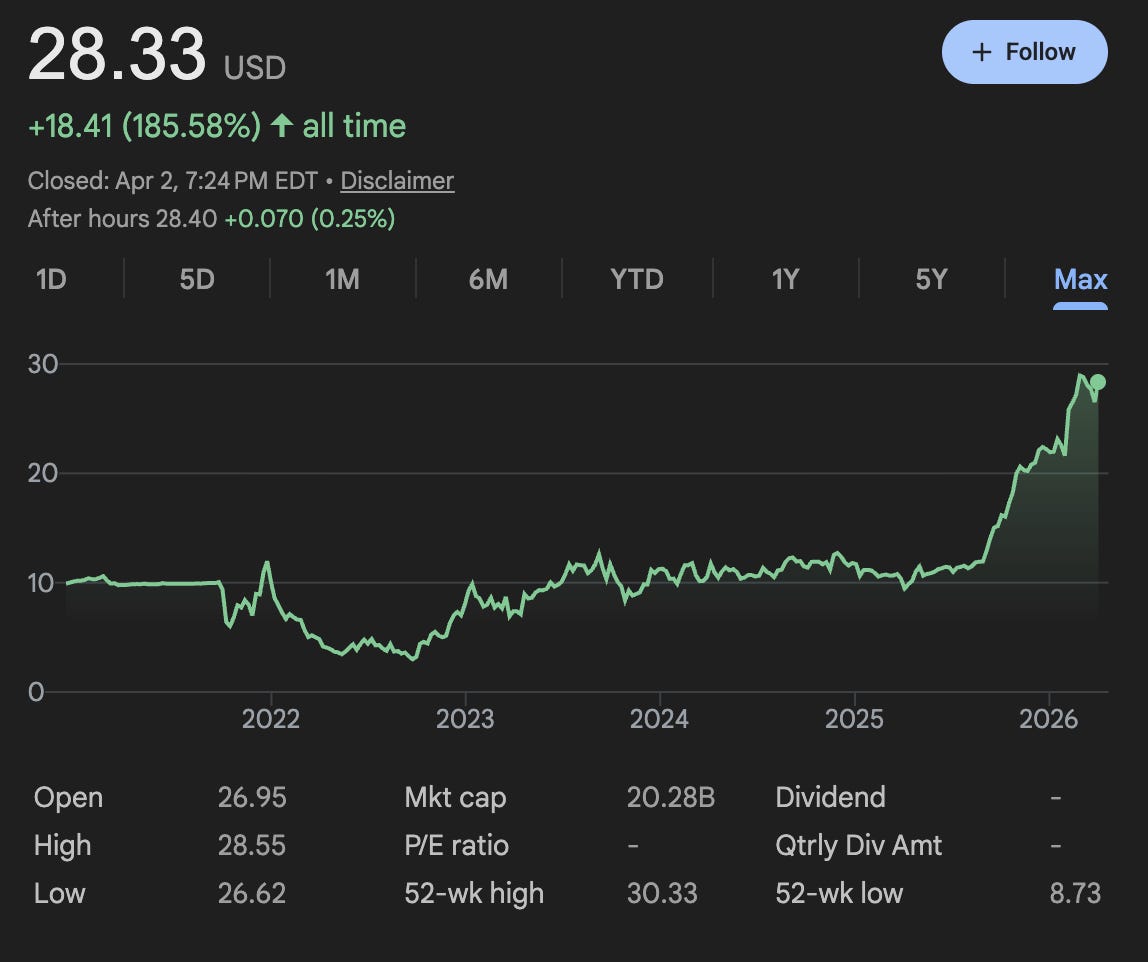

>The model did not work. Within eighteen months Centessa was shutting down spokes. By 2023, they had abandoned the hub-and-spoke model entirely and pivoted to a single-asset company focused on orexin agonists for sleep disorders. That pivot, to be clear, worked spectacularly.
To me this looks like the hub-and-spoke model working? It makes sense that most of the spokes would be shut down and they would invest in the successful spoke. Is the "not working" part the fact that they didn't keep making more spokes to replace the ones that didn't work?